Related article: Summary of research on aging and Olmesartan

Table of Contents
Aging (senescence)
The biological basis of aging (also known as senescence) is the subject of debate. A number of theories suggest aging is inevitable, the product of “wear and tear” or a result of some evolutionary necessity. For example, Skulachev argues that aging performs a specific biological function, with the turnover in new members of a species promoting greater evolutionary fitness.1) One of the longstanding problems of such theories is that they fail to account for dramatically different lifespans, or for the fact that chronic diseases are identical in presentation to diseases of the aging. We have a lot to learn about 'diseases of the aging'.
Recovery from some of the diseases of aging
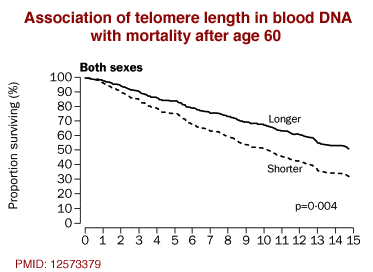
InflammationThe complex biological response of vascular tissues to harmful stimuli such as pathogens or damaged cells. It is a protective attempt by the organism to remove the injurious stimuli as well as initiate the healing process for the tissue., But Not Telomere Length, Predicts Successful Ageing at Extreme Old Age: A Longitudinal Study of Semi-supercentenarians. 2)
Health of the hypothalamus may be a controlling factor as demonstrated in a mouse study by Yalin Zhang et al
Mechanistically, hypothalamic stem/progenitor cells contributed greatly to exosomal microRNAs (miRNAs) in the cerebrospinal fluid, and these exosomal miRNAs declined during ageing, whereas central treatment with healthy hypothalamic stem/progenitor cell-secreted exosomes led to the slowing of ageing. Hypothalamic stem cells control ageing speed partly through exosomal miRNAs
and another with senior author Dongsheng Cai, M.D., Ph.D., (professor of molecular pharmacology at Einstein.)
The hypothalamus was known to regulate important processes including growth, development, reproduction and metabolism. In a 2013 Nature paper, Einstein researchers made the surprising finding that the hypothalamus also regulates aging throughout the body. Now, the scientists have pinpointed the cells in the hypothalamus that control aging: a tiny population of adult neural stem cells, which were known to be responsible for forming new brain neurons.
“Our research shows that the number of hypothalamic neural stem cells naturally declines over the life of the animal, and this decline accelerates aging,” says senior author Dongsheng Cai, M.D., Ph.D., (professor of molecular pharmacology at Einstein. “But we also found that the effects of this loss are not irreversible. By replenishing these stem cells or the molecules they produce, it's possible to slow and even reverse various aspects of aging throughout the body.” natural decline in neural stem cells over the life of the animal
According to the Marshall PathogenesisA description for how chronic inflammatory diseases originate and develop., chronic microbes drive the aging process, in light of the fact that diseases and symptoms of aging are at their essence, inflammatory conditions. It is the damage done by microbes coupled with the body's response to these pathogens which is responsible for aging skin, wrinkles, and the wear and tear on organs.
Conventional explanations for aging
A number of unsatisfactory theories have been touted to explain why aging occurs:
- Wear-and-tear theory – According to the wear-and-tear theory of aging, senescence is the result of chance. The human body is constantly wearing out and being repaired. Like components of an aging car, parts of the body wear out from repeated use.
- Aging-clock theory – Aging is programmed into our bodies like a body ticking away from the moment of conception. For example, female mammals are born with a finite number of egg cells in the ovaries that produce estrogen.
- Cross-linkage theory – Connective tissue in the body, such as the skin or the lens of the eye, loses elasticity with advancing age, allowing for the accumulation of harmful molecule.
- Free radicals – Free radicals are highly reactive and toxic when they come into contact with cell structures, thus generating biologically abnormal molecules. The articles pH Balancing and antioxidant supplementation address this theory.
- Cellular theory – According to this theory, the normal body has a finite potential to replicate and maintain functional capacity (Hayflick limit).
Breaking up defunct cells
Article in Press Senolytics in idiopathic pulmonary fibrosis
Selectively ablating senescent cells using dasatinib plus quercetin (DQ) alleviates IPF-related dysfunction in bleomycin-administered mice.
Our first-in-humans open-label pilot supports study feasibility and provides initial evidence that senolytics may alleviate physical dysfunction in IPF 3).
Diseases of the aging are chronic diseases
Moody writes, “Normal aging can be defined as an underlying time-dependent biological process that, although not itself a disease, involves functional loss and susceptibility to disease and death.4) Is aging inevitable?
There are two key problems with most conventional theories of aging. There is simply too much variability in symptoms of aging. The dramatic discrepancy lifespans suggests it's more than just chance which causes the symptoms of senescence.
For example, consider an 18-year old male who has constipation, brain fogThe loss of intellectual functions such as reasoning; memory loss; and other neurological abilities that is severe enough to interfere with daily functioning., and insomnia. Omit his age, and on paper, he sounds like an 80-year old. How are his symptoms qualitatively any different than his 80-year old counterpart's?
According to the Marshall Pathogenesis, diseases of the aging are simply late onset chronic diseases caused by infection.
Aging is a super-category. We’ve gradually lumped together more and more symptoms under the category of natural aging. Many of these symptoms are the same as those caused by diseases that surely have an infectious cause. In that sense, you could view much of what we now call aging as an incapacitating illness that leads to a decrease in function. We know that inflammation and the interaction of the immune system with pathogens can destroy tissue. So it’s not surprising that the tissues of a person who harbors a lot of pathogens would age earlier and alter their biological structure earlier in life. I do believe it is inevitable that people will eventually die of old age, but I suspect that this should generally happen when they are 80-100 years old. But we are increasingly seeing signs of aging-related diseases in people who are much younger.
Paul Ewald, Bacteriality interview
Infection and decline in immune function
A typical feature of aging is a chronic, low-grade inflammation characterized by a general increase in the production of pro-inflammatory cytokinesAny of various protein molecules secreted by cells of the immune system that serve to regulate the immune system. and inflammatory markers.5) There is even a term for it: inflammaging. According to Franchesci: “A large part of the aging phenotype, including immunosenescence, is explained by an imbalance between inflammatory and anti-inflammatory networks, which results in the low grade chronic pro-inflammatory status we proposed to call inflammaging.”6)
Aging deeply affects (or is affected by!) the human microbiotaThe bacterial community in the human body. Many species in the microbiota contribute to the development of chronic disease.'s homeostasis with the host's immune system:7) 8)
- autoimmuneA condition or disease thought to arise from an overactive immune response of the body against substances and tissues normally present in the body – As people age, their risk for developing an “autoimmune” condition also increases.9) The article on autoimmune conditions discusses why so-called “autoantibodies” are merely antibodies generated in response to pathogenic bacterial cells that have been destroyed as a result of an active immune response.
- macrophage function – Macrophages, which act as “pathogen sensors”, lose the ability to initiate an inflammatory response as people age.10) Microbes use a variety of methods to infect macrophages. For example, Neisseria meningitidis prevents macrophage apoptosis via genes encoding nitric oxide detoxification and a porin, PorB.11)
This theory is based on the fact that genes affecting host organism longevity are represented by subpopulations: genes of host eukaryotic cells, commensal microbiotaThe bacterial community which causes chronic diseases - one which almost certainly includes multiple species and bacterial forms., and non-living genetic elements. ……….. we propose that lifespan and aging are defined by the accumulation of alterations over all genes of macroorganism and microbiome and the non-living genetic elements associated with them. 12)
Could the chronic inflammation associated with aging be caused by pathogens? Given the crudeness of tools now used to measure microbes and the ubiquity of the human microbiota, this seems like a reasonable if not inevitable conclusion as the early studies have begun to suggest.
At present, a study on gut microbiota composition shows that three main modifications occur in faecal microbiota from old frail subjects: a 26-fold reduction in the number of lactobacilli (which stimulate immune functions and help the nutrient absorption), a 3-fold reduction in the number of bacteriodes (which digest polysaccarides, some species are opportunist pathogens) and a 7-fold increase in the number of enterobacteriacee (potentially pathogens)13). Differences in faecal microbiota were also found in a study on people of different countries and ages, including aged and long-lived people14).
E. Cevenini15)
Infections may drive age-related memory loss
- cognitive and functional decline often follow severe sepsis – The researchers focused their attention on 516 individuals who survived hospitalization for severe sepsis and 4517 who survived a nonsepsis hospitalization. The average age of survivors at hospitalization was 76.9 years. Following severe sepsis hospitalization, but not nonsepsis general hospitalization, there was a significant increase in the odds of developing both cognitive and physical dysfunction that persisted throughout the 8-year follow-up period, the researchers report.16)
- aging immune cells in animal models – The decline in brain function associated with disease and old age could be due to the decline in the function of immune cells, which is likely caused by infection. As described in New Scientist, prompted by studies suggesting immune responses can help repair the nervous system, Jonathan Kipnis and colleagues at the University of Virginia created mice that lack CD4 cells, a kind of T-cell. They found the mice performed extremely poorly in tasks involving learning and memory, but when they were injected with CD4 cells from healthy mice, their memories improved.17) Similarly, when he killed CD4 cells in healthy mice, their memory declined. Further animal studies by Kipnis and others show that learning new tasks triggers a mild stress response within the brain, which prompts CD4 cells to rally to the meninges, the membranes that surround the brain. Here, they release IL-4, which both switches off the stress response and tells brain cells called astrocytes to release brain-derived neurotrophic factor, a protein that enhances learning.18) Whether these animal studies are relevant to human learning and memory remains unclear, but there is some indirect evidence to support it. For example, many chemotherapy drugs suppress the immune system, which might explain why some people with cancer develop “chemobrain” - a term used to describe the cognitive problems and memory loss associated with chemotherapy. Sluggish immune cells might also explain why our brains slow down as we age. “The number one cell affected by ageing is the T-cell,” says Kipnis. “I'm not saying it's the only factor leading to age-related dementia, but it could definitely be one of them.”
Research into effects of Olmesartan
Role of vitamin D metabolism
The VDRThe Vitamin D Receptor. A nuclear receptor located throughout the body that plays a key role in the innate immune response. plays a plays a crucial, often under-appreciated, role in the innate immune responseThe body's first line of defense against intracellular and other pathogens. According to the Marshall Pathogenesis the innate immune system becomes disabled as patients develop chronic disease.. Further underscoring the potential role of microbes in chronic disease, there are several demonstrated connections between VDR dysfunction and premature aging.
- VDR knockout mice show signs of premature aging – VDR knockout mice (mice that have their Vitamin D Receptors removed) show signs of premature aging:
Overall, VDR KO mice showed several aging related phenotypes, including poorer survival, early alopecia, thickened skin, enlarged sebaceous glands and development of epidermal cysts…. Unlike the wildtype controls, VDR KO mice lose their ability to swim after 6 months of age. Expression of all the genes was lower in old VDR KO mice, but only NF-kappaBA protein that stimulates the release of inflammatory cytokines in response to infection, Fgf-23, p53 and IGF1R were significantly lower. Since the phenotype of aged VDR knockout mice is similar to mouse models with hypervitaminosis D(3), our study suggests that VDR genetic ablation promotes premature aging in mice, and that vitamin D(3) homeostasis regulates physiological aging.
T. Keisala et al.19)
- Vitamin D restriction reduces signs of aging in certain mice – The Klotho-insufficient (klotho) mouse exhibits a syndrome that resembles human aging including retarded growth, osteoporosis, atherosclerosis, ectopic calcification, immunological deficiency, skin and general organ atrophy, hypogonadism and short lifespan.20) This syndrome is mitigated – in one researcher's words, “rescued” – when mice have their vitamin D restricted.21) Interestingly, while murine research is a mainstay of the community of researchers and patients interested in longevity, this line of research has not led for calls to restrict vitamin D in the name of health.
Falls and Fractures
New Prevention Guidelines for Falls and Fractures
The major change is the downgrade of the vitamin D supplementation recommendation for preventing falls from being a B grade (recommended based on high certainty of moderate benefit or on moderate certainty of moderate to substantial benefit) to a D grade (recommended against based on moderate or high certainty of no benefit or that harms outweigh the benefit). For falls prevention, exercise remains a B grade recommendation, and multifactorial interventions remains a C grade (selectively offer) recommendation. Both falls prevention recommendations are consistent with a 2017 meta-analysis of falls prevention interventions.
Recommendations for fracture prevention have not changed, including insufficient evidence to make recommendations on calcium and vitamin D to prevent fractures in men and premenopausal and postmenopausal women.
Diet
Unlike current food pyramid recommendations, vigorous health in maturity may require far more daily fruit and vegetables; less carbohydrate, much less meat and no processed meat.
see Food and drink
Gray hair
Main article: Gray hair
Recent research
Cell membranes are primarily made up of two types of lipids — phospholipids and glycolipids. Inside cells, these lipids bind to a molecule called CD1d that transports them to the surface. Once there, phospholipids stimulate phospholipid-reactive T cells, and glycolipids stimulate a different type of T cell called iNKTs.
On their way to the cell’s surface, phospholipids more easily attach themselves to CD1d molecules, making it more difficult for glycolipids to attach to CD1d. Because of this, it is harder for glycolipids to make it to the surface of the cell. This means that iNKTs cannot be as easily stimulated by glycolipids.
Scientists believe iNKT cells are necessary because they appear to protect cells against the progression of certain cancers and autoimmune diseases. However, iNKT cells are extremely active and can cause alcoholic hepatitis or other types of liver diseases if they are overstimulated. The phospholipid’s ability to more easily bind to CD1d molecules than glycolipids keeps a balance between the two cell types and maintains homeostasis in the immune system. 22)
Read more
- Declining testosterone levels in men not part of normal aging, study finds – A 2012 study found that declining testosterone levels are not an inevitable part of the aging process, as many people think. Men who had declines in testosterone were more likely to be those who became obese, had stopped smoking or were depressed at either clinic visit.
[PMID: 9467841]
[PMID: 26629551] [PMCID: 4634197] [DOI: 10.1016/j.ebiom.2015.07.029]
[PMID: 30616998] [PMCID: 6412088] [DOI: 10.1016/j.ebiom.2018.12.052]
[PMID: 20388071] [DOI: 10.2174/138161210790883840]
[PMID: 17116321] [DOI: 10.1016/j.mad.2006.11.016]
[PMID: 22283774] [DOI: 10.2174/138161212799436593]
[PMID: 1822969] [DOI: 10.3109/08916939108997112]
[PMID: 15268749] [DOI: 10.1111/j.1474-9728.2004.00102.x]
[PMID: 16369030] [PMCID: 1346626] [DOI: 10.1128/IAI.74.1.729-733.2006]
[PMID: 29978435] [PMCID: 6208853] [DOI: 10.1007/s12064-018-0267-4]
[PMID: 16204576] [PMCID: 1265947] [DOI: 10.1128/AEM.71.10.6438-6442.2005]
[PMID: 16461645] [PMCID: 1392899] [DOI: 10.1128/AEM.72.2.1027-1033.2006]
[PMID: 20978262] [DOI: 10.1001/jama.2010.1546]
[PMID: 15141078] [PMCID: 419577] [DOI: 10.1073/pnas.0402268101]
[PMID: 20439540] [PMCID: 2867291] [DOI: 10.1084/jem.20091419]
[PMID: 19500727] [DOI: 10.1016/j.jsbmb.2009.03.007]
[PMID: 19444937] [DOI: 10.1016/j.jsbmb.2008.12.020]
[PMID: 21276773] [DOI: 10.1016/j.brainres.2011.01.056]
[PMID: 30508304] [PMCID: 6405219] [DOI: 10.1002/eji.201847717]
[PMID: 12573379] [DOI: 10.1016/S0140-6736(03)12384-7]