Related article: Health maintenance and Olmesartan

Table of Contents
Olmesartan (Benicar)
Olmesartan medoxomil is often referred by its brand name equivalent, Benicar. Patients on the Marshall ProtocolA curative medical treatment for chronic inflammatory disease. Based on the Marshall Pathogenesis. (MP) take 40mg of olmesartanMedication taken regularly by patients on the Marshall Protocol for its ability to activate the Vitamin D Receptor. Also known by the trade name Benicar. four to six times daily. There is no substitute for olmesartan.
Insurance coverage for olmesartan varies. Some patients obtain their olmesartan through online pharmacies.
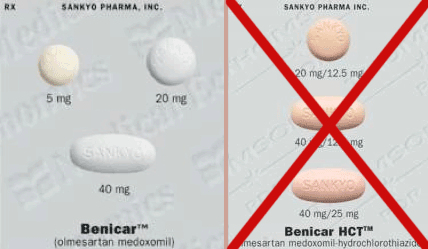
There is no substitute for olmesartan (Benicar)Medication taken regularly by patients on the Marshall Protocol for its ability to activate the Vitamin D Receptor.. Patients are warned not to order Benicar HCT, which contains a diuretic.
Olmesartan is both a potent anti-inflammatory and a Vitamin D Receptor agonistA substance such as olmesartan (Benicar) or 1,25-D which activates the Vitamin D Receptor and transcribes the genes necessary for a proper innate immune response., qualities which make the medication indispensable to patients on the Marshall Protocol (MP). Olmesartan has proven a safe therapeutic in a range of conditions.
Types of olmesartan
Brand name vs. generic olmesartan
Main article: Ordering olmesartan
Brand name olmesartan, which is manufactured by Daiichi Sankyo, Inc., is usually more expensive than generic olmesartan. The Benicar manufactured by Sankyo has an identification stamp on the tablet saying “C15” for the 40mg tablets or “C14” for the 20mg tablets. The stamp “Sankyo” is on the other side of these tablets.
Based on patient response, there is no evidence to indicate that Olmecip or any other generic is inferior. Olmecip smells differently, but this is due to it having a different binder formulation than the olmesartan made by Sankyo. Also, Olmecip dissolves more quickly, a benefit for MP patients when using frequent dosing.
Benicar HCT is contraindicated
Do not order Benicar HCT, also known as Benicar Hydrochlorothiazide. Thiazides are diuretics and contraindicated on the MP. Benicar HCT is orange. Benicar without HCT is white.
Ordering olmesartan
Main article: Ordering olmesartan
Dosing and administration of olmesartan
Main article: Dosing and administration of olmesartan
It is imperative that patients on the Marshall Protocol (MP) take regular doses of olmesartan medoxomil (the brand name of which is Benicar), a medication for which there is no substitute. Olmesartan has an highly safe track record. MP patients are required to take 40mg of olmesartan every six hours – that is, equally spaced doses, four times a day – with or without food. Patients who take olmesartan once a day are not doing the MP. Do not ramp or gradually increase olmesartan.
In cases where a dose is missed, patients may take an extra olmesartan as soon as possible and/or sublingually. When severe immunopathology strikes or in the case of an acute emergency, patients may increase the frequency of olmesartan to up to six times day. Another option is to take a dose of 60mg.
MP patients may want to keep at least one month of extra olmesartan on hand.
Patient response to olmesartan
Main article: Patient response to olmesartan
Related articles: Confronting uncertainty about the Marshall Protocol, Physicians' concerns and success rates for 5 Dx, Independent research into Olmesartan
Olmesartan (Benicar) has two actions. It palliates symptoms by reducing inflammationThe complex biological response of vascular tissues to harmful stimuli such as pathogens or damaged cells. It is a protective attempt by the organism to remove the injurious stimuli as well as initiate the healing process for the tissue., and it activates the innate immune responseThe body's first line of defense against intracellular and other pathogens. According to the Marshall Pathogenesis the innate immune system becomes disabled as patients develop chronic disease.. Patients who begin the Marshall Protocol (MP) may experience either, both, or neither. According to one rough estimate, 25% of patients who begin the MP experience immediate symptomatic relief when they begin the olmesartan blockade, about 25% feel no different, and the other 50% will experience some adjustment symptoms. Even for MP patients who have a minimal initial reaction to olmesartan, the medication almost always ultimately has strong antibacterial effects resulting in immunopathology. For olmesartan to be effective, it has to be dosed regularly: 40mg every 6-8 hours.
Keep an adequate emergency supply
The question of how much olmesartan (Benicar) to keep on hand requires consideration of a couple of factors. Patients who order their Benicar from overseas may want to plan for their medication to take up to a month to arrive. Delays in insurance coverage – for example, while awaiting appeal – can take even longer. Generally speaking, it helps to have at least one month of Benicar on hand.
In the event a patient runs out, it may be worth asking if one's doctor has Benicar samples as many do.
Also, patients who get their Benicar through insurance should refill their Benicar as soon as allowed. This will provide a patient with a few extra pills each refill.
Science behind olmesartan
Main article: Science behind olmesartan
Related article: Health maintenance and Olmesartan
Patients on the Marshall Protocol (MP) take olmesartan (Benicar), a drug whose actions are well known, every six hours. In general, olmesartan tends to be prescribed for its antihypertensive properties due to the fact that is an angiotensin receptor blocker. A growing body of research supports the use of olmesartan as a part of a curative therapy for chronic disease.
For the purposes of the MP, olmesartan has two primary actions: it reduces inflammation by blocking the Nuclear Factor-kappaB cytokine pathway and it is an agonist of the Vitamin D ReceptorA nuclear receptor located throughout the body that plays a key role in the innate immune response. (VDRThe Vitamin D Receptor. A nuclear receptor located throughout the body that plays a key role in the innate immune response.). As a VDR agonist, olmesartan uniquely activates the innate immune responseThe body's first line of defense against intracellular and other pathogens. According to the Marshall Pathogenesis the innate immune system becomes disabled as patients develop chronic disease.. Research supports the safety of the doses used by MP patients.
Olmesartan has minimal interactions with other drugs and is one of the safest drugs on the market.
Olmesartan and kidney disease
Main article: Olmesartan and kidney disease
As a matter of course, markers of kidney function including blood urea nitrogen (BUN) and creatinine will fluctuate while a patient is on the Marshall Protocol (MP).
There is a tendency for some physicians to become alarmed by these fluctuations, particularly if a patient has kidney disease. However, for the vast majority of patients these test results are an expected part of the healing process. In fact, a wide range of research shows that olmesartan is therapeutic for kidney disease.
Safety of olmesartan
Main article: Safety of olmesartan
Related article: Recent research into Celiac disease
Some patients and healthcare providers have expressed concerns about the safety of higher than typical doses of olmesartan (Benicar).
Ample research supports the fact that olmesartanMedication taken regularly by patients on the Marshall Protocol for its ability to activate the Vitamin D Receptor. Also known by the trade name Benicar. is one of the safest and has the most gentle side effects profiles of almost any drug on the market – as opposed to Benicar HCT, which contains a thiazide and is harmful.
I feel there are simply no adverse reactions or negative side effects that I need to worry about while taking the Marshall ProtocolA curative medical treatment for chronic inflammatory disease. Based on the Marshall Pathogenesis. medications. It is an extremely safe approach. Personally, I believe overtreating this condition is preferable than undertreating as the side effects of both antibiotics and Benicar are so minimal compared to the risk of [disease] recurrence.
Greg Blaney, M.D.
For those with gastric issues, it is recommended that the pill be crushed and mixed with applesauce or similar to reduce impact on gastric lining.