
Table of Contents
L-form bacteria
As a part of their natural life cycle, bacteria can transform into a variety of forms. One of those phases is the L-formDifficult-to-culture bacteria that lack a cell wall and are not detectable by traditional culturing processes. Sometimes referred to as cell wall deficient bacteria..
L-form bacteriaDifficult-to-culture bacteria that lack a cell wall and are not detectable by traditional culturing processes. Sometimes referred to as cell wall deficient bacteria., also known as cell wall deficient bacteria, are a phase of bacteria that are very small and lack cell walls.
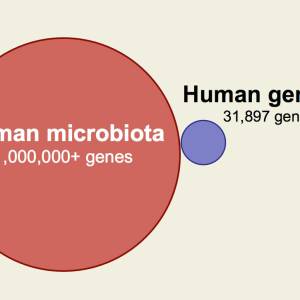
Though the subject of a great deal of research over the last 100 years and implicated in a variety of diseases, L-formsDifficult-to-culture bacteria that lack a cell wall and are not detectable by traditional culturing processes. Sometimes referred to as cell wall deficient bacteria. remain largely misunderstood - or at the very least, underappreciated - by the medical research community. According to the Marshall PathogenesisA description for how chronic inflammatory diseases originate and develop., L-forms are part of a metagenomic microbiotaThe community of bacterial pathogens including those in an intracellular and biofilm state which cause chronic disease. responsible for chronic disease.
Species capable of L-form transformation
Thus far, researchers have identified over 50 different species of bacteria capable of transforming into the L-form and it is likely that more species will be found in the coming years. “Probably most bacterial species can be converted into L-forms if treated with the antibiotics that inhibit cell wall synthesis,” states researcher Josep Casadesus. 1)
Some of the species of L-form bacteria that have been implicated in chronic disease include Bacillus anthracis, Treponema pallidum, Mycobacterium tuberculosis, Helicobacter pylori, Rickettsia prowazekii, and Borrelia burgdorferi. Not all species cause disease.
Size and shape
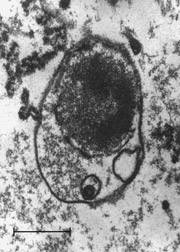
L-form bacteria are pleomorphic, that is, they can change size and shape. During much of their lifetimes they are tiny, about 0.01 microns in diameter.
Since they are smaller than viruses or fungal particles, they cannot be seen with a normal optical microscope. The small, individual forms of L-form bacteria are often referred to as coccoid bodies. Coccoid bodies sometimes group together, assuming the appearance of a string of pearls
Occasionally L-form bacteria break out of the cells. In the lab they can grow into long, thin biofilm A structured community of microorganisms encapsulated within a self-developed protective matrix and living together. filaments that can reach 60-70 microns in length. The biofilm filaments are composed of L-form bacteria and a protective protein sheath. For reasons still unknown, L-forms can also grow into large “giant” bodies.
L-form bacteria lack flagella, long slender appendages that allow some forms of bacteria to propel themselves forward by using a whip-like motion. Instead they glide to their destinations in a snail-like fashion.
Groups of L-form bacteria are often encased inside tubules. They are also separated from the environment inside the cell by a membrane or exoskeleton that keeps them from being digested by the cell.
Resistance to certain antibiotics
Cell wall deficient variants of bacteria cannot be killed by many commonly used antibiotics. Multiple studies, from many years ago2) to the present, have also shown that when one of the Beta-lactam antibiotics, a class of antibiotics that includes penicillin, are applied to wild-type bacteria in a Petri dish, small colonies of L-form bacteria form on the edges of the plate. According to Casadesus:3)
Treatment with penicillin does not merely select for L-forms (which are penicillin resistant) but actually induces L-form growth.
Josep Casadesus
Replication and reproduction
L-form bacteria replicate in various ways, including budding, filamentous growth and binary fission. Some species of L-forms such as Proteus can form large bodies that replicate by division. In other instances, granules bud from the body of the bacterium and give rise to small L-form colonies.
Survival mechanisms
Classical forms of most bacterial species can be found in the bloodstream. However L-form bacteria have figured out how to successfully infect and live inside the very cells of the immune system whose role is to kill bacteria. Once inside these cells, they can no longer be detected by the immune system and are able to persist in the body over long periods of time. L-form bacteria can infect many types of cells but prefer to infect white blood cells called macrophages.
Several very recent studies have confirmed the fact that bacteria can live inside the cells of the immune system. In a paper published in the Journal of Immunology by a team at the University of Michigan Medical School, Gabreil Nunez, senior author of the 2007 paper, stated “In our study, the presence of bacterial microbes inside the cell is what triggers the immune response.”4)
Similarly, a team of researchers at the Bacterienne Institute in France released a paper detailing how the bacteria E. coli is able to live inside the cells of the immune system.
[E. coli are] true invasive pathogens, able to invade intestinal epithelial cells and replicate intracellularly. Strains also survive and replicate within the macrophages.
Nathalie Rolhion et al. 5)
Markova has provided evidence that E. coli can survive lethal treatments such as boiling or autoclaving (subjecting equipment to high pressure steam at 121 °C or more) by transitioning into the L-form.6)
<html> <!–
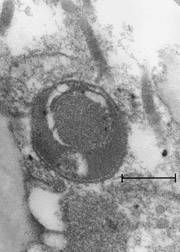
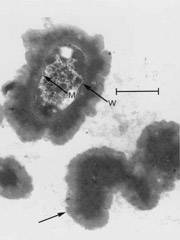
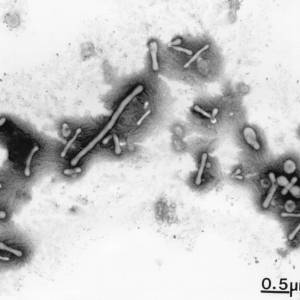
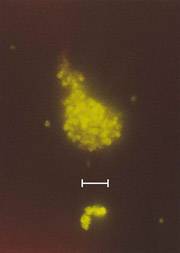
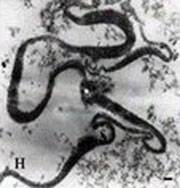
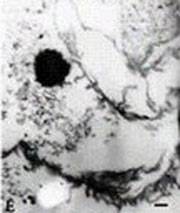
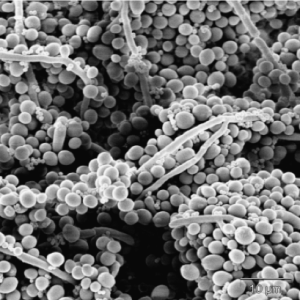
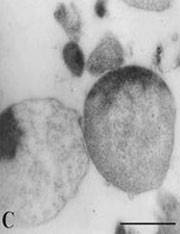
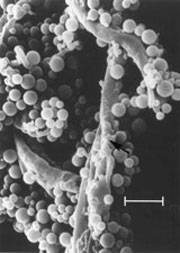
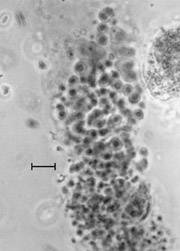
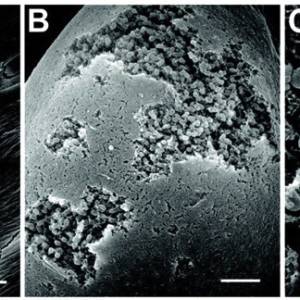
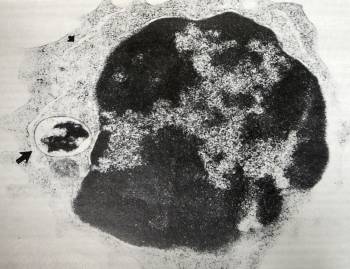
Images of L-forms
L-form bacteria have been photographed by a wide variety of researchers including Gerald Domingue 7) and Nadya Markova. 8)
Here are various images of L-forms. (Click for a larger image and description.)
–></html>
Culturing and detection
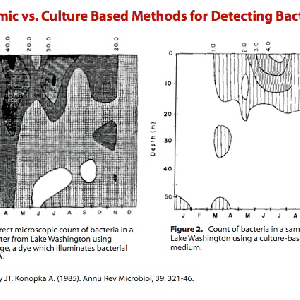
Once bacteria have transformed into the L-form they can no longer be detected by ordinary laboratory procedures. Although scientists have known about L-form bacteria for over a century, many of them have not been detected in tissue and blood samples because they are very difficult to culture.
Forms of bacteria with cell walls can be easily grown outside the body (grown in vitro). However L-form bacteria have great difficulty surviving in a foreign environment. In order to grow them successfully in the lab, conditions must be similar to those in the human body (grown in vivo). Consequently they can be cultured on a medium called blood agar at very specific temperatures and at a certain pH.
The concept that some bacteria cannot grow in vitroA technique of performing a given procedure in a controlled environment outside of a living organism - usually a laboratory. is not new. Scientists have known for decades that neither (Syphilis Treponema pallidum ) nor leprosy (Mycobacterium leprae) cannot be easily cultivated outside the body.
L-form bacteria take several measures to ensure they can survive for as long as possible inside a cell. They are able to infect all types of white blood cells, but seem to prefer to infect macrophages, the type of white blood cell, which at 45 days, has the longest life span.
Several studies have shown that once inside a macrophage, L-form bacteria are able to delay the process of apoptosis, or programmed cell death, allowing them to thrive inside the cell for a period of time even longer than 45 days.
Life without a wall or division machine in Bacillus subtilis. 9)
PCR testing
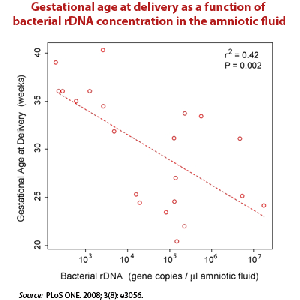
Classical bacterial forms can be detected by a lab test called Polymerase Chain Reaction (PCR). PCR identifies and amplifies the proteins and DNA of bacteria that have been killed. However since L-form bacteria are able to persist inside the macrophages for such extended periods of time, few of them die and only tiny amounts of L-form bacterial proteins and genetic material reach the bloodstream at any given time; an amount so small that the PCR test cannot pick them up.
Even if a few small fragments from L-forms that have been killed are identified by PCR testing, the remains are often not from the bacterial species causing the most harm to the patient. This is because the most well adapted, persistent bacterial species are the ones who have developed the most effective survival mechanisms and are consequently least likely to die.
Antibody testing
L-forms can also not be detected with antibody testing. Antibodies are Y-shaped proteins that are found in blood. They are used by the immune system to identify and neutralize foreign objects including bacteria.
However, antibodies only form in response to bacteria that have died. Since L-form bacteria are able to persist for such long periods of time inside the cells, very few antibodies are created in response to their presence.
Constant rediscovery
In 1915, Löhnis authored a paper entitled “Life cycles of the bacteria” in which they marveled at the complexity of the bacterial life cycle but also his failure to recognize its potential significance earlier:
The senior author also did not know that for more than 11 years he had in his collection, patiently waiting to be photographed, that fine prepárate now shown in figure 6 (PI. A) until, as stated before, he decided to take down his “theoretical blinders.” We have already mentioned that a careful study of the illustrations contained in our daily used textbooks will now reveal several things which we were so very well trained not to see. Certainly the German philosopher Lichtenberg made a very wise remark when he said: Was jederman für ausgemacht hält, verdient am meisten untersucht zu werden. [What everyone thinks identified deserves to be studied the most]
More recently, scientists such as Lida Mattman at Wayne State University, Gerald Domingue, and Nadya Markova have worked extensively with the L-form and figured out new ways to grow and view the pathogens. These techniques include a variety of special staining techniques. Nevertheless, many doctors and researchers still question whether the L-forms actually exist.
Mattman and others have spent decades figuring out how to correctly culture the L-form. Applying their techniques correctly requires rigorous adherence to specific guidelines. Mattman has said that, over and over again, researchers misinterpreted just one of the steps required to correctly grow the bacteria. They then report to the medical community that no L-forms appear in their samples.
Unfortunately, in the area of L-form or cell wall-defective bacteriology, too often there have been conclusions (anecdotal) drawn without supporting scientific data. In my opinion, many of these studies have hampered progress in the field and especially the role of these cryptic organisms in bacterial persistence and expression of disease.
Gerald Domingue, professor emeritus, Tulane University School of Medicine
“Features of a number of important but poorly explained human clinical syndromes strongly indicate a microbial etiology,” states Relman. “In these syndromes, the failure of cultivation-dependent microbial detection methods reveals our ignorance of microbial growth requirements.” 10)
Select diseases linked to l-forms
- psoriasis – In a 2009 study, Wang investigated the carriage rate of cell wall deficient bacteria in the tonsil or pharynx of psoriasis patients. CWDB were isolated from 74.2% of psoriasis patients, 23.5% of chronic tonsillitis patients and only 6.3% of controls. Meanwhile, our study showed that CWDB and wide-type bacteria significantly enhanced the production of IFN-gamma, in vitro, and peripheral blood mononuclear cells proliferation.11)
Koch's postulates
Another problem with fully understanding the role of L-forms rests with the fact that many researchers rely on a series of rules called Koch’s postulates when interpreting research data. The postulates state that only one pathogen can cause a given disease. But research has shown each chronic disease is the result of infection with multiple species of L-forms.
This means that separate teams of researchers often detect different L-forms in patients with the same disease. For example both Borrelia burgdorferi and Rickettsia helvetica have been detected in patients with sarcoidosis. These findings make little sense to researchers still bent on adhering to Koch’s postulates.
Infection with L-form bacteria
People are exposed to L-form bacteria in many places. Because they cannot be killed by pasteurization or chlorination, L-form bacteria can be found in milk, food, and water. They can be transmitted via sperm, intimate contact, and can be passed from mother to child during childbirth. Since they are too small to be filtered during the purification processes used in pharmaceutical manufacturing procedures, they can be transmitted through injectable medicines. They have even been cultured from dry soil.
One 2007 trial for an AIDS vaccine had to be abruptly discontinued when scientists realized that the vaccine somehow raised the risk of infection. The researchers administering the trial stressed that the vaccine could not itself cause the infection. Although it was not mentioned in the news story about the incident, one very plausible hypothesis is that the vaccines were infected with L-form bacteria.
Once macrophages and other cells have been infected with L-form bacteria, the bacteria circulate in the blood and tissues. In some cases they cluster together in clumps called granulomas. In other cases, they accumulate in regions such as the joints.
After L-form bacteria have successfully invaded a cell, they begin to use the nutrients inside the cell to their own advantage, disturbing the cell’s delicate chemical balance. They are also able to take control of the host’s genetic material, which allows them to create proteins that enhance their ability to survive.
Inflammation as evidence of infection
L-form bacteria cause inflammationThe complex biological response of vascular tissues to harmful stimuli such as pathogens or damaged cells. It is a protective attempt by the organism to remove the injurious stimuli as well as initiate the healing process for the tissue. and painful symptoms by taking control of the protein known as Nuclear Factor Kappa B. They are able to activate proteins that increase the activity of Nuclear Factor Kappa B, which subsequently moves to the nucleus or center of the cell. Once there, it turns on a variety of genes that cause the release of inflammatory cytokinesAny of various protein molecules secreted by cells of the immune system that serve to regulate the immune system., proteins that generate pain and/or fatigue. These cytokines include interferon gamma and TNF alpha.
In this way, an inflammatory response is correlated with diseases caused by L-form bacteria.
An inflammatory immune response—one of the body’s primary means to protect against infection—defines multiple established infectious causes of chronic diseases, including some cancers.
David Relman, MD
Inflammation also drives many chronic conditions that are still classified as (noninfectious) autoimmuneA condition or disease thought to arise from an overactive immune response of the body against substances and tissues normally present in the body or immune-mediated (e.g., systemic lupus erythematosus, rheumatoid arthritis, Crohn’s disease). Both [the innate and adaptive immune systems] play critical roles in the pathogenesis of these inflammatory syndromes. Therefore, inflammation is a clear potential link between infectious agents and chronic diseases.
Siobhán M. O'Connor of the Centers for Disease Control and Prevention concurs, stating, “The epidemiologic, clinical, and pathologic features of many chronic inflammatory diseases are consistent with a microbial cause.”
Recent research
L-form bacteria cohabitants in human blood: significance for health and diseases. 12)
Possible role of L-form switching in recurrent urinary tract infection 13)
Future research
There seems to be little incentive for scientists to study the L-form. Since the bacteria can be killed by simple low-dose antibiotic therapy, drug companies have little interest in investing money into related research. Researchers studying the L-form often find themselves with very little grant money but must still work long, tedious hours in the lab.
It is generally agreed among scientists that L-form bacteria are extraordinarily intriguing, interesting tools for biological study, yet the most neglected area of research has been on the role of these organisms in disease, particularly in host-pathogen interactions.
Gerald Domingue, PhD
Directed evolution to shape the function of enzymes, has received the Nobel Prize in Chemistry 2018.
[PMID: 18008373] [DOI: 10.1002/bies.20680]
[PMID: 13475673] [DOI: 10.1099/00221287-17-1-64]
[PMID: 17433728] [DOI: 10.1016/j.immuni.2007.03.008]
[PMID: 17476674] [DOI: 10.1002/ibd.20176]
[PMID: 9105757] [PMCID: 172922] [DOI: 10.1128/CMR.10.2.320]
[PMID: 10959728] [DOI: 10.1016/S1438-4221(00)80123-X]
[PMID: 19212404] [DOI: 10.1038/nature07742]
[PMID: 9716951] [PMCID: 2640285] [DOI: 10.3201/eid0403.980310]
[PMID: 20137493]
[PMID: 28715646]
[PMID: 31558767] [PMCID: 6763468] [DOI: 10.1038/s41467-019-12359-3]
[PMID: 19806199] [PMCID: 2752164] [DOI: 10.1371/journal.pone.0007316]

