Related article: Palliative vs. curative treatments

Table of Contents
Edit the page and hit Save. See Formatting Syntax for Wiki syntax. Please edit the page only if you can improve it. If you want to test some things, learn to make your first steps on the playground.
Corticosteroids
For many physicians, corticosteroidsA first-line treatment for a number of diseases. Corticosteroids work by slowing the innate immune response. This provides some patients with temporary symptom palliation but exacerbates the disease over the long-term by allowing chronic pathogens to proliferate., of which glucocorticoids are one kind, are a first-line treatment for a number of diseases, including multiple sclerosis, sarcoidosis, lupus, and rheumatoid arthritis. Corticosteroids suppress the innate immune responseThe body's first line of defense against intracellular and other pathogens. According to the Marshall Pathogenesis the innate immune system becomes disabled as patients develop chronic disease., which provides some patients with temporary symptom palliation but also allows chronic pathogens to proliferate.
Research shows that any kind of short-term symptomatic improvement from corticosteroidA first-line treatment for a number of diseases. Corticosteroids work by slowing the innate immune response. This provides some patients with temporary symptom palliation but exacerbates the disease over the long-term by allowing chronic pathogens to proliferate. use does not last, and that over the longer term, use of the drugs can have adverse consequences, including a litany of side effects. For even short periods of time, steroid use can become genuinely addictive. For their own safety, patients on the Marshall ProtocolA curative medical treatment for chronic inflammatory disease. Based on the Marshall Pathogenesis. (MP) must wean off of them as opposed to discontinuing them outright.
Types and varieties of corticosteroids
Ther term corticosteroids refers to a family of drugs that includes synthetic drugs, such as prednisolone, and natural ones such as cortisol, an adrenal hormone which is found naturally in the body. Though synthetic and natural corticosteroids are both potent anti-inflammatory compounds, the synthetics exert a stronger effect.
Corticosteroids include mineralocorticoids, which affect salt and water retention, and glucocorticoids, which affect protein, fat, and carbohydrate metabolism.
The following are common varieties of corticosteroids. Note that the substance names are in lower case and that any brand names are capitalized:
- beclomethasone (Beconase)
- betamethasone
- cortisol - has an equivalency to prednisone of approximately 1:2
- cortisone
- Deflazacort
- dexamethasone (Decadron)
- DHEA
- fluticazone (Flonase)
- fludrocortisone (Florinef)
- hydrocortisone (Cortef)
- methylprednisolone (Medrol, Medrol DosePak, Solu-Medrol, Solu-Medrol infusion)
- prednisolone (Prednisone) - a Vitamin D ReceptorA nuclear receptor located throughout the body that plays a key role in the innate immune response. (VDRThe Vitamin D Receptor. A nuclear receptor located throughout the body that plays a key role in the innate immune response.) antagonist, which down-regulates the actions of a protein dimer called NuclearFactor-kappaB, a protein essential to the immune response and vital for balance of bone generation.
- triamcinolone
Methods of administration
Corticosteroids can be administered by injection, topically, nasally, orally, or via eye drops. MP patients are strongly discouraged from using any of these corticosteroid forms because they are absorbed systemically and are, to some degree, immunosuppressive.
Weaning from corticosteroids
Main article: Weaning from corticosteroids
Corticosteroids are immunosuppressive
Interesting to compare LDN with Pred. Basically both medicines suppress proinflammatory response to LPS(endotoxin) though through different pathways(assuming the research on Naltrexone and Pred is correct of course).
The effects of steroids on the outcome of sepsis are dose dependent. Low doses appear to be beneficial, but high doses do not improve outcome for reasons that are insufficiently understood. The effects of steroids on systemic inflammationThe complex biological response of vascular tissues to harmful stimuli such as pathogens or damaged cells. It is a protective attempt by the organism to remove the injurious stimuli as well as initiate the healing process for the tissue. as a function of dose have not previously been studied in humans.
These data indicate that prednisolone dose-dependently and differentially influences the systemic activation of different host response pathways during human endotoxemia. de Kruif et al 1)
A sudden and unprecedented increase in low dose naltrexone (LDN) prescribing in Norway. Patient and prescriber characteristics, and dispense patterns. A drug utilization cohort study by Raknes G. and Småbrekke L.
We emphasize that we have neither examined indications, nor the efficacy or safety of LDN use. Many patients became persistent users of LDN, and this may be interpreted as a perceived acceptable effect and safety profile among these patients. This cannot be considered as a proof of LDN treatment efficacy for any condition. 2)
Parkitny L. and Younger J found that LDN was associated with reduced plasma concentrations of interleukin (IL)-1β, IL-1Ra, IL-2, IL-4, IL-5, IL-6, IL-10, IL-12p40, IL-12p70, IL-15, IL-17A, IL-27, interferon (IFN)-α, transforming growth factor (TGF)-α, TGF-β, tumor necrosis factor (TNF)-α, and granulocyte-colony stimulating factor (G-CSF). We also found a 15% reduction of FM-associated pain and an 18% reduction in overall symptoms. 3)
Short-term palliative effect
Main article: Palliative vs. curative treatments
Patients with chronic disease may find that while taking corticosteroids, both symptoms and markers of the disease, including X-rays, liver enzymes, etc., may temporarily decrease. This is due to the short-lived decrease in inflammation as well as the fact that pathogens are not being killed. The bacteria multiply in the tissues without any hindrance once the corticosteroids have shut down the body's immune reaction.
Even if the dose of corticosteroids is continually increased over time, the sheer amount of toxins that the bacteria generate will, at some point, become uncontrollable.
Modulation of nuclear receptors
Corticosteroids act on a host of nuclear receptorsIntracellular receptor proteins that bind to hydrophobic signal molecules (such as steroid and thyroid hormones) or intracellular metabolites and are thus activated to bind to specific DNA sequences which affect transcription., activating some and inactivating others. For example, cortisol has an affinity for the glucocorticoid receptor, and aldoesterone has an affinity for the mineralocorticoid receptor. Corticosteroids also bind to other key receptors including the Vitamin D Receptor, PPAR-alpha, PPAR-gamma, and others.
Whether nuclear receptors are active or inactive is important because they control, and, when modulated, can upset everything from immune response to energy metabolism.
The function of the glucocorticoid receptor (GCR) is not fully known, as mice who are bred without it do not survive gestation.
Natural hormones are no different
The body exquisitely balances the endogenous production of its steroid hormones in a manner which cannot be mimicked with endogenous drugs. The problem with viewing cortisol or any other hormone as a supplement is that it acts as a steroid rather than a replacement.
Any dose of steroid which alters the body's actions - i.e., makes you feel better - is by its very nature unsafe in the long term…. By way of example, hydrocortisone alters operation of the VDR, thyroid-alpha, thyroid-beta, glucocorticoid, mineralcorticoid and too many other alterations for me to detail.
Trevor Marshall, PhD
Previous use of steroids' effect on recovery
MP patients who have previously used corticosteroids should be optimistic about recovery on the MP, but they are at somewhat of a disadvantage, because:
- Fibrosis, which is not stopped by steroids, encases bacterial pathogens in collagen, from which they will eventually begin to break free.
- The long-term use of steroids promotes parasitization of the more dense organs, rather than just soft-tissue in the lungs.
Corticosteroids are ineffective
There are no studies which show that steroids improve long-term prognosis in the treatment of illness. Van den Bosch and Grutters write4), “Remarkably, despite over 50 years of use, there is no proof of long-term (survival) benefit from corticosteroid treatment.”
The 2003 NIH ACCESS study5), the largest and most ambitious to date, validates that these drugs are ineffective for sarcoidosis patients, even for a time period as short as two years.
Table 3 in the paper shows how, for the majority of patients, markers of the disease process remained unchanged.
| FVC | FEV1 | CXR | Dyspnea | |
|---|---|---|---|---|
| Improved | 20.5% (44) | 21.9% (47) | 37.7% (81) | 19.5% (42) |
| Unchanged | 57.7% (124) | 56.7% (122) | 41.4% (89) | 67.0% (144) |
| Worse | 11.6% (25) | 11.2% (24) | 16.3% (35) | 13.5% (29) |
Other findings of the study:
- “Fifty of the 215 subjects (23%) developed one or more new organs involved with sarcoidosis over the two-year follow-up period.”
- Corticosteroids are not a curative therapy: “The use of corticosteroids may promote relapse of sarcoidosis when the medication is discontinued.”
- Any improvement “was not statistically related to whether patients did or did not receive therapy.”
- “End-stage pulmonary sarcoidosis usually develops over one or two decades.”
Latent pathogens grow slowly and accumulate over the course of decades. There is every reason to believe that a longer-term study as well as one looking at dose would further demonstrate how corticosteroids are worse than ineffective – they're dangerous. A more comprehensive paper would also have examined the range of negative side effects associated with coriticosteroid use.
A systematic review6) of studies looking at the efficacy of inhaled and oral corticosteroids for pulmonary sarcoidosis concluded:
Oral steroids improved the chest X-ray and a global score of CXR, symptoms and spirometry over 3-24 months. However, there is little evidence of an improvement in lung function. There are limited data beyond two years to indicate whether oral steroids have any modifying effect on long-term disease progression.
Corticosteroids cause relapse
According to a comparative study7), 74% of patients relapse after trying to ease up their Prednisone dose, a significantly higher percentage of the time, and:
Relapse… rarely occurred in patients who had not been treated with corticosteroids in the past. The striking difference in relapse rate between treated and untreated patients suggests that patients with disease that would later be severe and protracted were almost unerringly identified early in their course.
Jonathan E. Gottlieb et al.
Also:
Corticosteroids contribute to the prolongation of the disease by delaying resolution.
Side effects of corticosteroid therapy
Corticosteroid therapy can produce a range of negative side effects in humans. Side effects tend to be dose- and duration-dependent. The table below summarizes some of the more common side effects. A more complete and up-to-date list of such studies is available at PubMed.
| System or function | Side effect |
|---|---|
| brain | loss of brain tissue8) loss of cognition9) |
| bone | avascular necrosis of the bone10) 11) osteoporosis12) osteonecrosis13) |
| endocrine | suppression of adrenal response14) 15) widespread endocrine dysfunction16) Cushing's Syndrome17) |
| eyes | cataracts18) glaucoma19) 20) range of other side effects21) |
| gastrointestinal | gastrointestinal hemorrhage22) |
| muscle | loss of skeletal muscle23) myopathy (muscle weakness)24) 25) 26) |
| psychological | dementia27) |
| sexual, female | menstruation disturbance28) |
| sexual, male | decrease testosterone29) 30) |
| skin | skin atrophy31) |
Side effects in children
Whether taking injectable or inhaled corticosteroids, children are particularly susceptible to the negative long-term effects of the drugs. The table below summarizes some of the more common side effects in children. A more complete and up-to-date list of such studies is available at PubMed.
| System or function | Side effect |
|---|---|
| bone | higher risk of fractures32) loss of density33) |
| dental | eruption of permanent molars34) |
| endocrine | suppression of adrenal response35) growth suppression36) 37) 38) Cushing's syndrome39) |
| immunological | allergies40) |
| psychological | psychosis41) poor attention42) |
| skin | skin death43) |
| urinary | kidney stones44) |
Other side effects from corticosteroids include:
- increased risk of infection
- fluid retention and increase in weight
- diabetes
- indigestion or worsening of peptic ulcer
- changes in mood
- impaired healing
- stretch marks
- skin thinning
- bruising
- increased facial hair
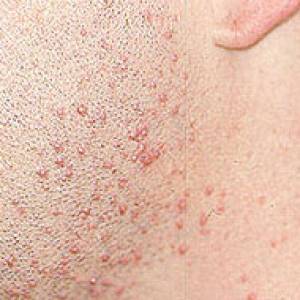
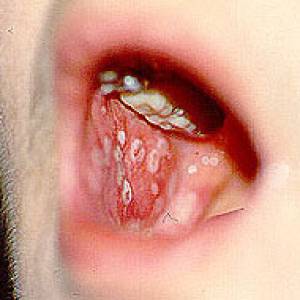
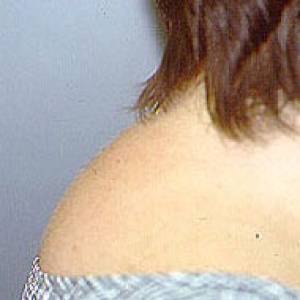
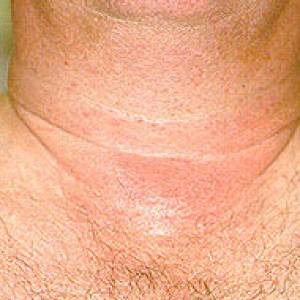
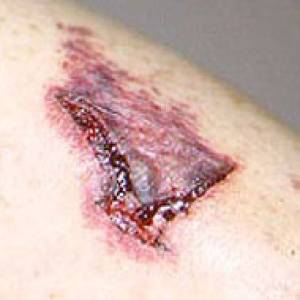
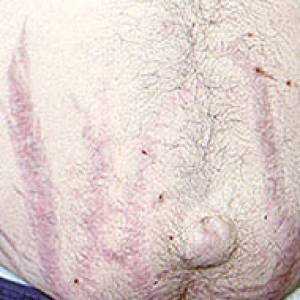
Images of side effects
Courtesy the Johns Hopkins Vasculitis Center, these images are of corticosteroid users. (Click for a larger image and description.)
Working with a conventionally-minded doctor
Most physicians are well-accustomed to using corticosteroids to treat chronic disease. MP patients who are being treated with corticosteroids should ask themselves these questions if their health care practitioners are reluctant to change that treatment:
- Am I suffering treatment-induced diseases or damage as a result of taking high-dose steroids?
- Am I familiar with reports indicating that corticosteroids do not halt the advancement of sarcoidosis?
- Do I realize that withdrawal from corticosteroids causes relapse of disease symptoms that may be worse than the original symptoms?
Limited use of steroids for intolerable symptoms
While on the MP, limited use of sniffed, inhaled or topical steroids is allowed for intolerable symptoms of nasal congestion, shortness of breath or itching if management of MP medications fails to dampen immunopathologyA temporary increase in disease symptoms experienced by Marshall Protocol patients that results from the release of cytokines and endotoxins as disease-causing bacteria are killed. symptoms. Steroid eye drops may be necessary.
MP patients should consult with their health care practitioners to devise a plan to substitute other medication for symptom relief, if possible, or to discontinue the use of the steroid product as soon as the acute inflammation is under control.
[PMID: 17237435] [DOI: 10.4049/jimmunol.178.3.1845]
[PMID: 27670755] [PMCID: 5298009] [DOI: 10.1002/pds.4110]
[PMID: 28536359] [PMCID: 5489802] [DOI: 10.3390/biomedicines5020016]
[PMID: 16946094] [DOI: 10.1183/09031936.06.00105805]
[PMID: 14620163]
[PMID: 15846612] [PMCID: 6464973] [DOI: 10.1002/14651858.CD001114.pub2]
[PMID: 9118698] [DOI: 10.1378/chest.111.3.623]
[PMID: 15882880] [DOI: 10.1016/j.jns.2005.03.006]
[PMID: 12013026] [DOI: 10.1515/revneuro.2002.13.1.59]
[PMID: 11850924]
[PMID: 10099819] [DOI: 10.1155/1999/517893]
[PMID: 15732231] [DOI: 10.1007/BF02914518]
[PMID: 18757967] [DOI: 10.1302/0301-620X.90B9.20056]
[PMID: 10683005] [DOI: 10.1016/S0140-6736(99)06290-X]
[PMID: 9623166]
[PMID: 2320881]
[PMID: 15850848] [DOI: 10.1016/j.ecl.2005.01.013]
[PMID: 3541262] [DOI: 10.1016/0039-6257(86)90077-9]
[PMID: 10641955] [DOI: 10.2165/00002512-199915060-00004]
[PMID: 7701998]
[PMID: 11820911] [DOI: 10.2165/00002018-200225010-00004]
[PMID: 8927965]
[PMID: 6851197] [DOI: 10.1111/j.1365-2265.1983.tb03198.x]
[PMID: 9551512]
[PMID: 15677408] [DOI: 10.1196/annals.1314.018]
[PMID: 2815743]
[PMID: 3083749] [DOI: 10.7326/0003-4819-104-5-648]
[PMID: 7983641]
[PMID: 11393207] [DOI: 10.1034/j.1600-0846.2001.70203.x]
[PMID: 12733732] [DOI: 10.1359/jbmr.2003.18.5.913]
[PMID: 18595975] [PMCID: 2928657] [DOI: 10.1542/peds.2007-3381]
[PMID: 15470830]
[PMID: 15269067] [PMCID: 1720050] [DOI: 10.1136/adc.2002.022533])((. Inhaled steroids and the risk of adrenal suppression in children. Expert Opin Drug Saf. 2002 Sep;1(3):237-44. doi: 10.1517/14740338.1.3.237.
[PMID: 12904139] [DOI: 10.1517/14740338.1.3.237]
[PMID: 1823076] [DOI: 10.1159/000182157]
[PMID: 9709954] [DOI: 10.1210/jcem.83.8.5036]
[PMID: 11826253]
[PMID: 18183788] [DOI: 10.1515/jpem.2007.20.11.1173]
[PMID: 11456363]
[PMID: 15851433] [PMCID: 1720409] [DOI: 10.1136/adc.2003.041541]
[PMID: 14577022] [DOI: 10.1007/s00467-003-1295-x]
[PMID: 1806315] [DOI: 10.1111/j.1365-2230.1991.tb01225.x]
[PMID: 8576746]
[PMID: 20008700] [DOI: 10.1001/archinternmed.2009.384]
[PMID: 16172857] [PMCID: 7088345] [DOI: 10.1007/s10096-005-0004-z]